A population screening programme is a systematic offer of medical examinations to screen for a specific disease among a population of apparently healthy, asymptomatic individuals.
Aim and target group
The aim of the cervical cancer screening programme is to reduce mortality by detecting early stages of the disease. A precancerous stage is usually easy to treat, which can prevent cervical cancer from developing.
People between the ages of 30 and 60 are invited to take part in the cervical cancer screening programme. The target group of the programme is women living in the Netherlands and transgender persons or people with an X/O gender registration and having a uterus.
Primary process
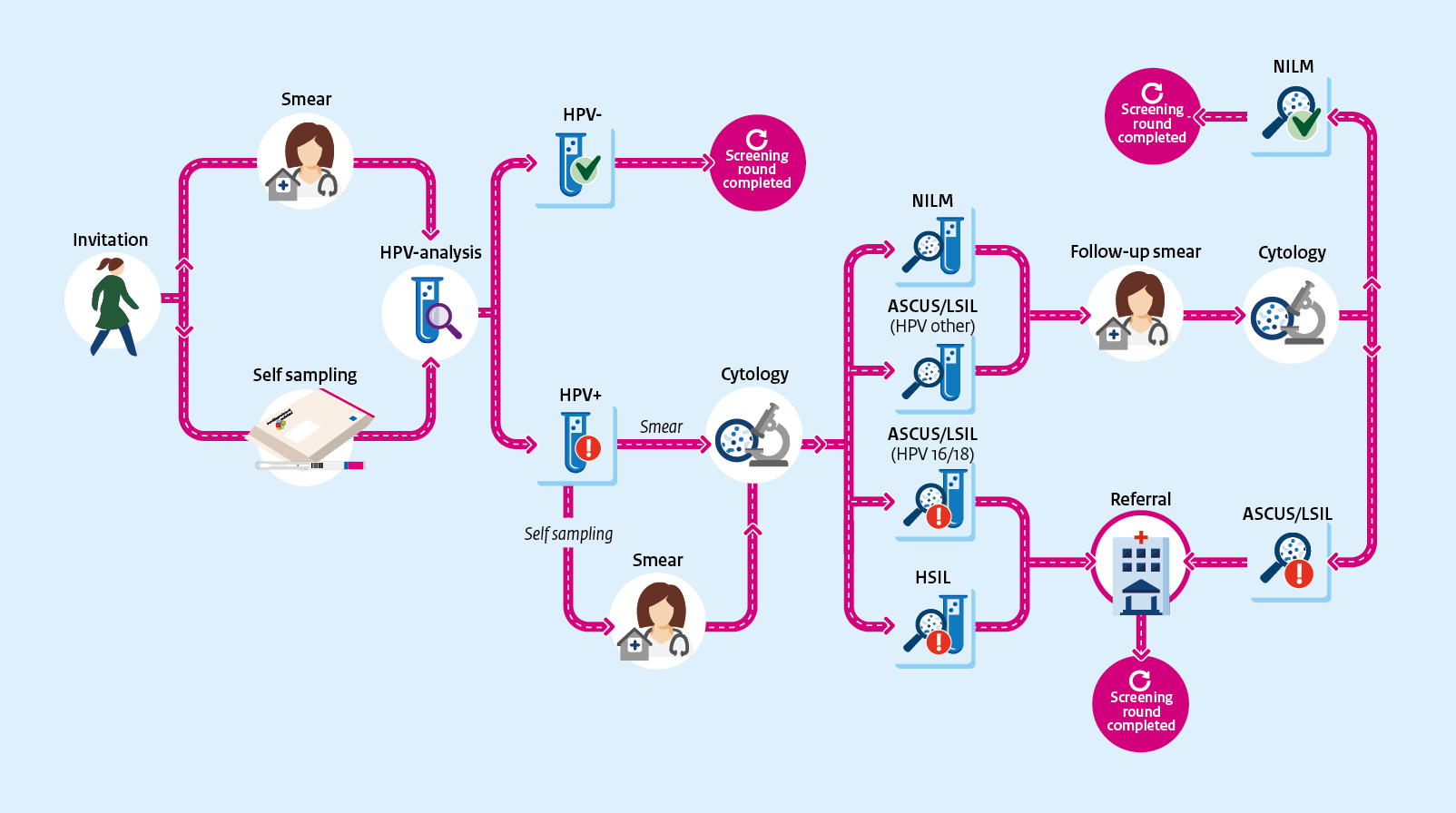
The primary cervical cancer screening process is shown here in diagram form.
View the image as a digitally accessible pdf
Overview
The cervical cancer screening starts with the selection of people who are eligible to be invited. The screening ends by informing the participants of the results. Participants who, based on the results, require further diagnosis and possibly treatment are referred to regular healthcare providers.
Select
The screening organisation selects people who are eligible for an invitation based on their age. Data from the Key Register of Persons (BRP) are used for this. Persons who have cancelled or who are not eligible based on a previous result are not selected. Transgender persons or people with an X/O gender registration and a uterus can also take part. They have to register with the screening organisation once.
Invite
The screening organisation sends the invitations for the population screening according to an established invitation schedule. People who are eligible for an invitation receive it around their birthday, together with the invitation leaflet.
In addition, there are other letters and invitations:
• 30-year-olds receive a pre-announcement of the invitation 6 weeks before the first invitation. They will then receive a specific invitation letter. This invitation includes a self-sampling kit.
• People who have not responded to the invitation will receive a reminder letter after 12 weeks. A self-sampling kit is then included.
• In the event of a postponement due to pregnancy, the invitee will receive a new invitation letter 6 weeks after the pregnancy.
Screen
Invitees first choose whether or not to participate. They can participate in two ways: at home with the self-sampling kit or with a smear test at the general practice.
The screening test by means of a smear test starts when the invitee makes an appointment at the general practice to have a smear taken. The smear is tested in one of the three screening laboratories for the presence of hrHPV (high risk Human Papillomavirus). If HPV is found, a cytological assessment is carried out to determine whether there are also abnormal cells. This is done on the same smear in the same laboratory.
The screening test by means of self-sampling starts when someone requests the self-sampling kit from the screening organisation. Invitees aged 30 and those aged 35-60 who have not responded to the invitations will receive the self-sampling kit automatically. They can use this to collect vaginal material themselves and then send this to be tested for hrHPV.. The self-sampling kit is tested for hrHPV. If the participant tests positive for HPV, a smear test is still needed for a cytological evaluation.
Communicate result
In the case of a smear test, both the GP and the participant are informed of the result and the accompanying advice. The results of a self-sampling kit are not communicated to the GP. The participant receives the results of the smear test or the self-sampling kit from the screening organisation. HPV-positive participants receive the result letter and the result leaflet. If the result is HPV-positive with abnormal cells (HSIL), the GP will try to inform the participant before the participant receives the result letter.
Refer
HPV-positive participants with cytological abnormalities will be advised to undergo further examination by a gynaecologist. They will receive a referral from their GP according to an established referral schedule. The gynaecologist's examination is not part of the screening programme.